Most Trusted eQMS on the ServiceNow Platform

Discover the benefits of a cloud-native eQMS on ServiceNow – agile, compliant, and integrated. Quality Forward is your trusted ServiceNow eQMS expert.
ISO 9001:2026 – What Quality Leaders Need to Know Ahead

Prepare for ISO 9001:2026 with a modern eQMS. Learn key changes, timeline, and how to future-proof your QMS. Stay compliant and competitive today.
7 Steps for a Smooth Move from Legacy to Cloud QMS

Migrating from a legacy system to a Cloud QMS? Discover 7 proven steps to ensure a smooth QMS migration and unlock the benefits of a cloud-based quality system.
Top AI QMS Solutions for Regulated Industries

Discover the best AI QMS solutions for regulated industries. Compare AI-powered and AI-based QMS software to boost compliance, efficiency, and ROI.
Quality Management System (QMS) For Clinical Trials

Learn how a Quality Management System (QMS) ensures compliance, data integrity, and audit readiness in clinical trials.
FDA Audit Checklist: Is Your Manufacturing QA Team Ready?

Use this FDA audit checklist to ensure your manufacturing QA team stays inspection-ready. Master FDA audit prep with a strong quality system.
GMP vs. cGMP: A Compliance Guide for QA Teams

Discover the key difference between GMP and cGMP and why “current” matters. Learn how to stay compliant with modern cGMP standards in pharma and medtech.
QA Guide: Best CAPA Management Software

Learn about the best Corrective and Preventive Action (CAPA) Software to automate compliance, reduce risk, and resolve issues faster across FDA and ISO-regulated industries
Complete Guide to GxP: Compliance, QMS & Best Practices

Learn what GxP Compliance means, how it differs from GMP, and discover key compliance practices, ALCOA principles, and QMS solutions for regulated industries.
Electronic QMS vs. Paper QMS: What Saves More Time/Money?

Electronic QMS vs. Paper QMS: Discover which system saves more time and money while boosting compliance, efficiency, and scalability in regulated industries.
Mastering the 7 Principles for ISO 9001:2015

Discover how quality control in pharmaceuticals ensures safety, compliance, and efficiency with modern QMS tools and regulatory standards.
Best Practices for Quality Control in Pharmaceuticals

Discover how quality control in pharmaceuticals ensures safety, compliance, and efficiency with modern QMS tools and regulatory standards.
QMS 101: Mastering CAPA Management in Pharmaceuticals

Learn how to master CAPA management in pharmaceuticals with this essential QMS guide. Improve compliance, reduce risks, and streamline quality control.
FDA 483 vs. Warning Letter: Compliance Guide

Learn how to prevent and respond to an FDA 483 Warning Letter with expert guidance, CAPA strategies, and eQMS tools for regulatory compliance.
QMS 101: How to Implement a Quality Management System

Learn how to successfully run a Quality Management System (QMS) implementation with our step-by-step guide.
eQMS in Manufacturing: How to Cut Audit Time by 50%

Learn how to implement an eQMS in manufacturing environments to improve quality, ensure compliance, and streamline operations across your entire workflow.
QMS 101: Risk Management in Quality Assurance Guide

Explore how risk management enhances quality assurance in life sciences. Learn best practices, ISO/FDA compliance, and how to automate risk workflows in eQMS.
Top QMS Standards You Need to Know (ISO, FDA, EU)

Explore key QMS standards like ISO 9001, ISO 13485, and FDA 21 CFR Part 820. Learn how digital QMS platforms help you meet global compliance requirements.
Ultimate Guide to ISO 9001 Compliance in Life Sciences

Explore how to meet ISO 9001 requirements in pharma, biotech, and medtech. Learn key clauses, documentation needs, and how eQMS supports certification.
How to Simplify Audit Management in Life Sciences

Discover how audit management systems help pharma, biotech, and medtech companies stay compliant. Automate audit trails, track CAPAs, and stay inspection-ready.
QMS 101: Change Control Software for Life Sciences
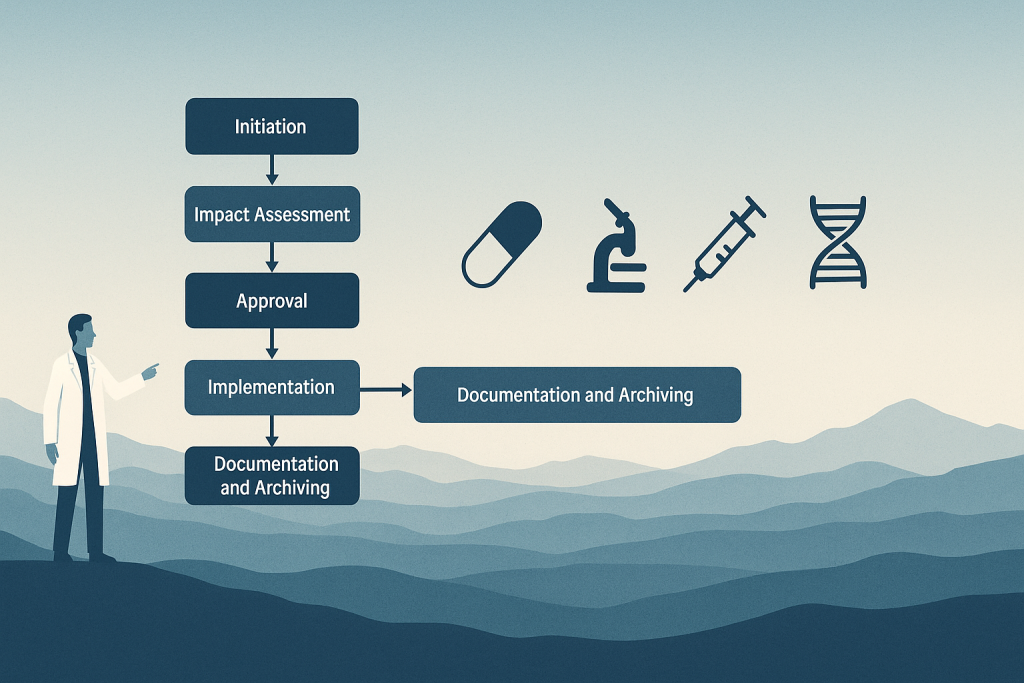
Learn change control management with confidence in pharma, medtech, and biotech. Learn best practices, regulatory compliance, and how to streamline it with eQMS.
QMS 101: Guide to Safety Incident Response in Life Sciences

Learn about Safety Incident Management in Life Sciecnes; pharma, biotech, and medtech. Discover best practices, compliance tips, and how eQMS streamlines reporting.
QMS 101: Understanding Supplier Qualification in Life Sciences

Learn the key steps in supplier qualification for life sciences, including regulatory requirements, risk management, and CAPA integration for compliance.
What are Corrective and Preventive Actions (CAPA)

Learn everything about Corrective and Preventive Actions (CAPA), its role in quality management, and compliance with FDA, ISO, and GMP regulations. Improve your QMS today!
FDA 21 CFR Part 11: A Complete Compliance Checklist Guide

Ensure FDA compliance with 21 CFR Part 11 for electronic records & signatures in pharmaceuticals, biotech, and medical devices. Learn key requirements, validation tips, and best practices for regulatory success.
What is Good Manufacturing Practice (GMP) in Life Sciences

Ensure Good Manufacturing Practice (GMP) compliance with key regulations, best practices, and industry standards for pharmaceuticals, medical devices, and food safety.
Why You Need a Document Management Quality System

Improve compliance and efficiency with a Document Management Quality System. Discover best practices, automation tips, and key features for seamless document control.
ICH Q10: Everything You Need for Pharma Quality Systems

Discover the essentials of ICH Q10 and its impact on pharmaceutical quality systems. Learn how to implement this guideline for compliance and efficiency.
Guide to Quality Management System (QMS)

Learn everything about Quality Management Systems (QMS), their benefits, and how they ensure compliance in pharmaceuticals, biotech, and medical devices. Discover best practices and industry standards like GMP, ISO 9001, and FDA 21 CFR Part 11.
